 
Lesser energy is required for the extraction of low reactive metals or metals present at the bottom of the reactivity series, as compared to those above hydrogen gas.The lesser stable oxide or compound that a metal forms, the easier it is for it to be extracted.Thermal decomposition is the name for the method of extraction of low reactive metals like Hg, Ag, Pt, and Au, which can be reduced to metals by heating alone, which is a mild phenomenon.The three least reactive metals, silver, platinum, and gold, are freely present in nature.Due to the un-reactive nature of these metals, their oxides can be reduced to metals by heating alone. 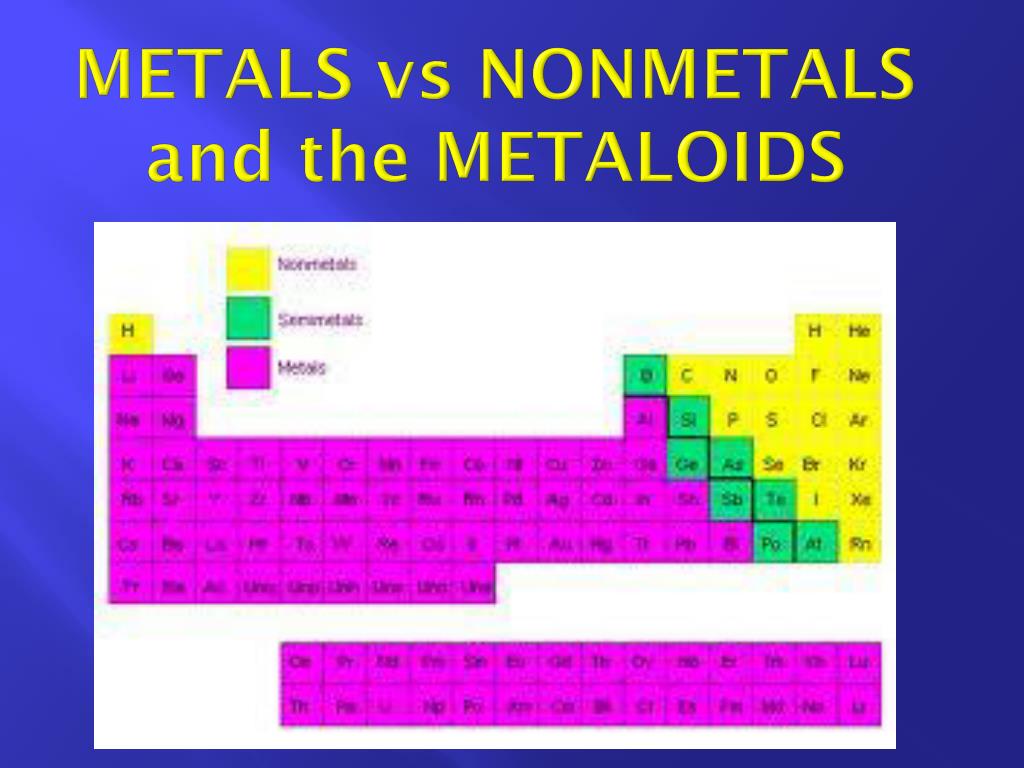
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
